How to Set Up a Compliance Document Portal That Passes Audits
A compliance document portal centralizes the collection, organization, and auditing of regulatory and policy documents. This guide covers the features your portal needs, how to structure it for different compliance frameworks, and how to keep it audit-ready year-round.

What Is a Compliance Document Portal?
A compliance document portal is a centralized platform for collecting, organizing, and auditing regulatory and policy documents with version tracking, expiration alerts, and audit-ready reporting.
That definition matters because most organizations don't have a compliance document problem. They have a compliance document location problem. Policies live in SharePoint. Certificates sit in someone's email. Training records are in the HR system. Vendor agreements are in a shared drive somewhere. When an auditor asks for evidence, someone spends two weeks pulling it all together.
The portal solves this by giving compliance teams a single place where every document has a clear owner, a known expiration date, and a full access history. Auditors get a controlled view of exactly what they need, nothing more.
This is different from a generic document management system. A DMS handles storage and retrieval for all business documents. A compliance portal adds the regulatory layer: version control tied to policy revisions, automated alerts when certifications expire, audit trails that show who accessed what and when, and structured workflows for document review and approval.
It's also different from a full GRC (Governance, Risk, and Compliance) platform. GRC tools like ServiceNow, Archer, or LogicGate handle risk assessments, control mapping, incident management, and compliance scoring. A compliance document portal focuses specifically on the document collection and evidence management layer, which is often the most time-consuming part of compliance work.
According to a Ponemon Institute study, the average cost of non-compliance is $14.82 million annually, roughly 2.71 times what organizations spend on compliance programs themselves. Much of that cost comes from failed audits, regulatory fines, and the operational disruption of scrambling to produce evidence after the fact.
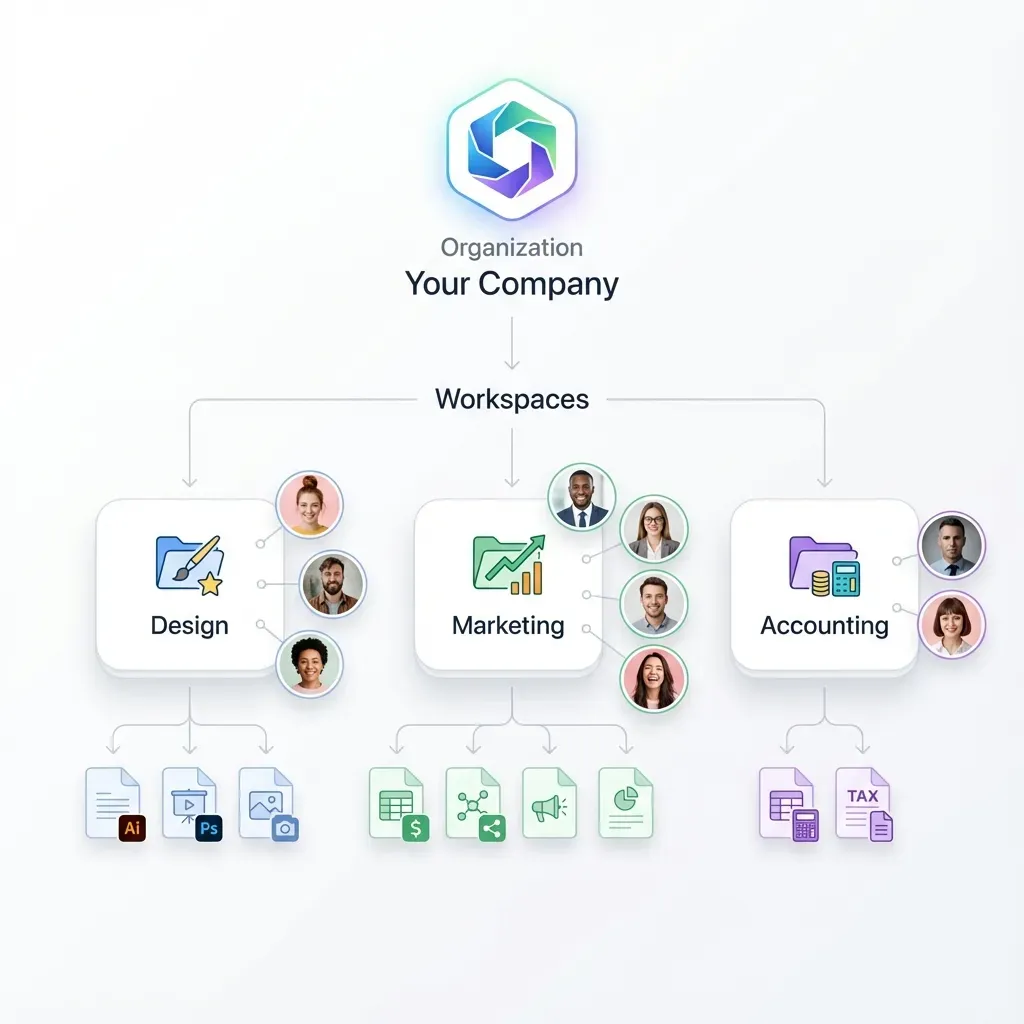
Features Every Compliance Portal Needs
Not every feature matters equally. Here's what separates a portal that passes audits from one that creates more work than it saves.
Version control with full history. Compliance documents change frequently. Policies get updated, certificates get renewed, training records accumulate. Your portal needs to track every version of every document, who changed it, when, and why. When an auditor asks "what was your data retention policy on March 15th?", you need an answer in seconds, not hours.
Expiration tracking and automated alerts. Certifications expire. Insurance policies lapse. Training requirements have renewal dates. A compliance portal should track expiration dates for every document type and send alerts 30, 60, and 90 days before deadlines. The alternative is a spreadsheet that someone forgets to update.
Granular access controls. Different stakeholders need different access levels. Internal compliance teams need full read/write access. Department heads need access to their area's documents. External auditors need read-only access to specific evidence packages. Guest users, like vendors submitting their own compliance documents, need upload access without seeing anything else.
Audit trail and activity logging. Every action in the portal should be logged: uploads, downloads, views, edits, permission changes, and login attempts. This isn't optional. Regulations like SOX, HIPAA, and GDPR explicitly require demonstrable access controls and activity records.
Document collection workflows. Compliance isn't just about storing your own documents. You need to collect documents from others: vendor certifications, employee training completions, third-party audit reports. The portal needs inbound collection workflows where external parties can upload documents into designated folders without accessing the rest of the system.
Search and retrieval. When an auditor requests "all vendor security assessments from the last 12 months," you need to find them fast. Full-text search, metadata filtering by date range, document type, department, and framework, and structured folder hierarchies all matter.
Checklist of must-have features:
- Automatic version numbering with change tracking
- Configurable expiration alerts (30/60/90 days)
- Role-based access with guest/auditor modes
- Immutable audit logs (who, what, when)
- Inbound document collection from external parties
- Full-text and metadata search
- Bulk export for audit packages
- Password-protected external access
- Retention policy enforcement
How to Structure Your Portal for Different Frameworks
The way you organize your compliance portal depends on which frameworks you're working with. A company preparing for SOC 2 has different document needs than one managing GDPR data subject requests.
SOC 2. Organize around the five Trust Services Criteria: security, availability, processing integrity, confidentiality, and privacy. Each criterion gets its own folder with subfolders for policies, evidence, and control documentation. For example, under Security, you'd have your information security policy, access control procedures, incident response plan, penetration testing reports, and employee security training records.
HIPAA. Structure around administrative safeguards, physical safeguards, and technical safeguards. Administrative safeguards include risk assessments, workforce training records, and business associate agreements. Physical safeguards cover facility access logs and workstation policies. Technical safeguards include encryption policies, access control documentation, and audit logs.
GDPR. Organize by obligation type: data processing records (Article 30), data protection impact assessments, consent records, data subject access request logs, breach notification procedures, and data processing agreements with third parties.
ISO 27001. Follow the Annex A control structure. Create folders for each control domain (A.5 through A.18) with subfolders for the Statement of Applicability, risk treatment plan, and evidence for each control.
Multi-framework environments. Most organizations deal with more than one framework. The practical approach is to create a primary folder structure around your most demanding framework, then use tags or metadata to map documents to additional frameworks. A single access control policy might satisfy SOC 2 CC6.1, ISO 27001 A.9, and HIPAA Technical Safeguards simultaneously. Cross-referencing prevents duplicate documents and the version drift that comes with them.
Regardless of framework, every compliance portal should have a top-level folder for cross-cutting documents: organizational charts, business continuity plans, vendor management policies, and employee handbooks. These documents get referenced across multiple frameworks and shouldn't be buried in a single category.

Build an Audit-Ready Document Portal
Centralize your compliance documents with branded portals, granular permissions, and full audit trails. Collect evidence from vendors, control auditor access, and search everything instantly.
Setting Up a Compliance Portal Step by Step
Here's a practical setup process that works regardless of which tool you choose.
Step 1: Inventory your existing documents
Before building anything, catalog what you already have and where it lives. Common locations include shared drives, email attachments, HR systems, legal folders, and individual employees' desktops. Create a spreadsheet listing every compliance document, its current location, owner, last update date, and expiration date (if applicable).
This step usually reveals two things: you have more documents than you thought, and many of them are outdated.
Step 2: Define your folder structure
Based on your compliance frameworks (see the section above), create a folder hierarchy. Keep it as flat as possible. Three levels deep is a good maximum: Framework > Category > Document Type. Deeper nesting makes documents harder to find and harder to maintain.
Step 3: Set up access roles
Define who needs access to what. Common roles include:
- Compliance admin: Full access, manages structure and permissions
- Department owner: Read/write access to their department's documents
- Internal reviewer: Read access to documents they need to review or approve
- External auditor: Time-limited read-only access to specific evidence packages
- Vendor/partner: Upload-only access to submit their compliance documents
Step 4: Migrate and organize
Move documents into the portal in batches, starting with the framework you'll be audited on next. As you migrate, update metadata: set expiration dates, assign owners, tag documents by framework, and archive outdated versions.
Step 5: Set up collection workflows
For documents you need from others, create inbound collection points. Vendors should be able to submit insurance certificates, security questionnaires, and compliance attestations through a branded upload form, not by emailing PDFs to your compliance team.
Step 6: Configure alerts and reviews
Set expiration alerts for every document that has a renewal date. Schedule quarterly reviews for all policies and annual reviews for the overall portal structure. Assign each review to a specific person, not "the compliance team."
Step 7: Test with a mock audit
Before your next real audit, run a mock one. Ask someone outside the compliance team to request specific documents. Time how long it takes to produce them. If it takes more than a few minutes per request, your portal structure needs work.
Choosing the Right Tool
Compliance portal tools range from repurposed file storage to specialized GRC platforms. The right choice depends on your compliance complexity and budget.
Repurposed file sharing (Google Drive, SharePoint, Dropbox). These work for very early-stage compliance programs. You can create a folder structure, set basic permissions, and share links. The limitations show up fast: no expiration tracking, weak audit trails, no inbound collection workflows, and no way to give auditors a branded, controlled view of evidence. If you're managing compliance across more than one framework or handling external audits regularly, you'll outgrow this quickly.
Dedicated compliance portals. Tools built specifically for compliance document management include dedicated portals, evidence management platforms, and workspace solutions with compliance-ready features. These give you version control, audit trails, access controls, and collection workflows in one place.
Fast.io works well for compliance teams that need to collect documents from external parties, maintain audit trails, and give auditors controlled access. Its Content Portals provide branded, password-protected spaces where you control exactly what each viewer sees. Granular permissions at the org, workspace, folder, and file level let you mirror your compliance framework structure. The built-in audit trail logs every file operation, membership change, and access event. Receive shares let vendors and partners upload compliance documents directly into designated workspaces without accessing anything else.
For teams already using AI in their workflows, Fast.io's Intelligence Mode auto-indexes uploaded documents, letting you search compliance files by meaning rather than just filename. Ask "show me all vendor security assessments that mention data encryption" and get results with citations to specific pages.
Full GRC platforms (ServiceNow GRC, Archer, LogicGate, Hyperproof). These handle the entire governance, risk, and compliance lifecycle: risk assessments, control mapping, automated evidence collection, continuous monitoring, and compliance scoring. They're powerful but complex, with implementation timelines measured in months and costs in the tens of thousands annually. If you need risk quantification and continuous compliance monitoring across dozens of frameworks, a GRC platform makes sense. If your primary need is organizing documents and passing audits, it's overkill.
The practical middle ground for most organizations: use a dedicated compliance portal for document management and evidence collection, and layer a GRC platform on top only when your compliance program outgrows document-centric workflows.
Keeping Your Portal Audit-Ready Year-Round
Setting up a compliance portal is the easy part. Keeping it useful requires ongoing discipline.
Assign document owners, not teams. Every document in the portal should have a single named owner responsible for keeping it current. "The compliance team" isn't an owner. Unassigned documents are the ones that go stale.
Schedule automated reviews. Policies should be reviewed at least annually. Certificates and licenses need review before expiration. Training records need updates after each training cycle. Build these review cycles into the portal's alert system rather than relying on calendar reminders.
Run quarterly access audits. Review who has access to the portal and whether they still need it. Remove former employees, former vendors, and auditors whose engagement has ended. Stale access is a finding in almost every compliance audit.
Maintain a change log. When policies change, document what changed and why. A version history shows the what, but auditors often want the why: regulatory update, incident response, organizational restructuring, or periodic review.
Practice evidence retrieval. The worst time to discover your portal is disorganized is during an actual audit. Run tabletop exercises quarterly: pick a random audit request from a past audit and time how long it takes to produce the evidence. Target under five minutes per request.
Keep collection workflows active. Vendor compliance documents expire, too. Set up recurring collection cycles for insurance certificates, security questionnaires, and compliance attestations. Automate reminders so vendors know when their documents are due for renewal.
Companies that manage compliance manually spend roughly three times more effort on audits than those with structured systems, according to compliance operations benchmarks. The portal doesn't eliminate the work, but it compresses the audit preparation window from weeks to days.
Frequently Asked Questions
What is a compliance document portal?
A compliance document portal is a centralized platform for collecting, organizing, and auditing regulatory and policy documents. It provides version tracking, expiration alerts, granular access controls, and audit-ready reporting so compliance teams can produce evidence quickly during audits.
How do you manage compliance documents?
Start by inventorying all compliance documents across your organization and consolidating them in a central portal. Assign an owner to each document, set expiration alerts for certifications and policies, define access roles for internal teams and external auditors, and schedule regular reviews. Use collection workflows to gather documents from vendors and partners.
What documents are needed for compliance?
The specific documents depend on your regulatory framework. Common requirements include security policies, access control procedures, risk assessments, business continuity plans, employee training records, vendor agreements, incident response plans, data processing records, and audit reports. Most frameworks also require evidence of regular policy reviews and updates.
How do you set up a compliance portal?
Inventory your existing compliance documents, define a folder structure based on your regulatory frameworks, set up access roles for different stakeholders, migrate documents in batches, configure expiration alerts, create collection workflows for external parties, and test the setup with a mock audit before your next real one.
What's the difference between a compliance portal and a GRC platform?
A compliance portal focuses on document collection, organization, and evidence management. A GRC platform handles the broader compliance lifecycle including risk assessments, control mapping, continuous monitoring, and compliance scoring. Many organizations start with a document portal and add GRC capabilities as their program matures.
How often should compliance documents be reviewed?
Policies should be reviewed at least annually, though some regulations require more frequent reviews. Certifications and licenses need review before their expiration dates. Access controls should be audited quarterly. Training records should be updated after each training cycle. Building automated review reminders into your portal prevents documents from going stale.
Related Resources

Build an Audit-Ready Document Portal
Centralize your compliance documents with branded portals, granular permissions, and full audit trails. Collect evidence from vendors, control auditor access, and search everything instantly.
